
The modern FDA approval maze is becoming less a straight corridor and more a high-speed roundabout—and investors riding with Eupraxia Pharmaceuticals (EPRX), Eli Lilly (LLY) and Merck (MRK) may find that’s exactly where outsized opportunity lives.
From Wall Poster to Wall Street
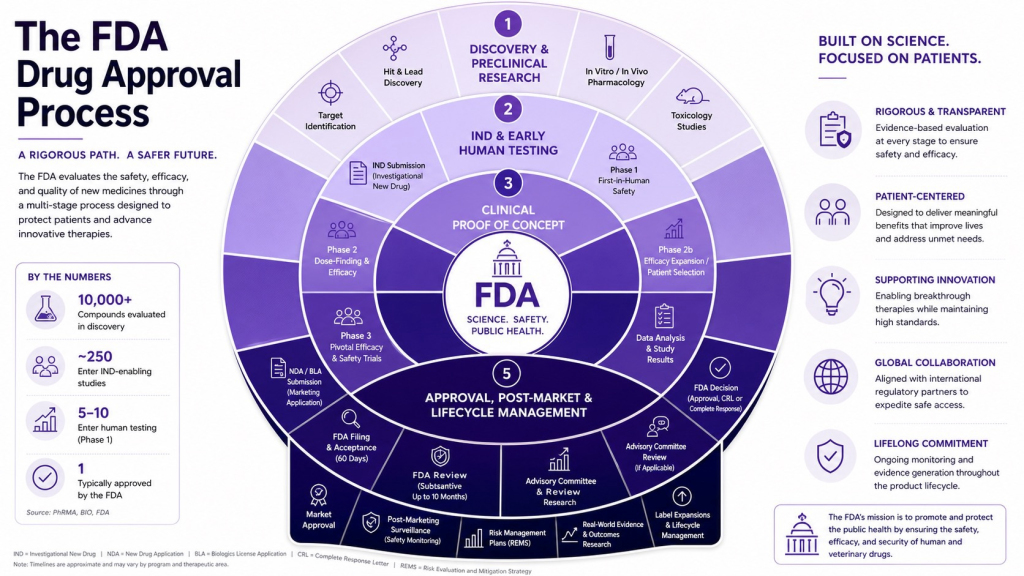
The classic FDA flowchart—discovery, clinical proof of concept, pivotal trials, approval, and post‑market monitoring—looks tidy enough pinned to a clinic wall. In practice, it now resembles air‑traffic control for an industry trying to land first-in-class obesity pills, precision-delivery platforms, and next‑generation cancer drugs on the same runway. For investors, the key isn’t memorizing every acronym on the graphic; it’s understanding how select companies are learning to bend the timeline without breaking the rules.
Eupraxia Pharmaceuticals, Eli Lilly, and Merck each offer a different angle on this new regulatory reality: one rewiring drug delivery, one redefining metabolic disease, and one defending oncology’s high ground. Together, they illustrate how the FDA’s evolving playbook is rewriting the risk‑reward calculus across the sector.
Eupraxia: Turning Old Drugs into New Stories
Eupraxia Pharmaceuticals (NASDAQ: EPRX) is not trying to out‑GLP‑1 Eli Lilly or out‑oncology Merck; instead, it is treating the FDA process itself as a design constraint—and a competitive weapon. The company’s core Diffusphere platform is built to deliver existing or well‑known drugs in a more precise, extended‑release manner, targeting local tissue over long durations while limiting systemic exposure. In a world where regulators increasingly like innovation but love safety databases, “better delivery of familiar agents” is more than a scientific nuance—it can be a regulatory shortcut.
Eupraxia has already seen the FDA nod in that direction: its EP‑104IAR candidate for knee osteoarthritis received Fast Track designation, opening the door to more frequent FDA interactions and potentially accelerated review if the data line up. Fast Track doesn’t lower the bar for approval, but it can shorten the distance between promising data and commercial reality—an important distinction for small caps whose most expensive line item is time. More recently, Eupraxia has been generating encouraging Phase 1b/2 data in eosinophilic esophagitis (EoE) with EP‑104GI, including 36‑week tissue health and symptom improvements that it is taking to the Digestive Disease Week (DDW) stage in Chicago.
For investors, the appeal is that Eupraxia is not betting the farm on exotic biology; it is using known pharmacology and aiming to let formulation engineering do the heavy lifting. If regulators continue to favor programs that pair innovation with risk containment, platforms like Diffusphere could become valuable partners or acquisition targets for larger players that prefer their pipeline upgrades with a side of de‑risking.
Eli Lilly: Fast Track to the Waistline and the Bottom Line
If Eupraxia is playing the long game with controlled delivery, Eli Lilly is running a full‑court press on cardiometabolic disease. The company’s GLP‑1 franchise has already reshaped revenue expectations across Big Pharma, and the pipeline suggests Lilly is nowhere near done. Orforglipron, its once‑daily oral GLP‑1, has now secured an FDA approval for obesity and weight-related conditions—part of a new expedited review initiative that cut the review to roughly 50 days.
This is not just another pill; it’s the regulatory equivalent of express checkout in a market projected to reach roughly 150 billion dollars in obesity drug sales by 2035, driven largely by GLP‑1 receptor agonists. Late‑stage trials showed double‑digit percentage weight loss at higher doses compared to low single‑digit declines with placebo, giving payers and physicians reason to take the drug seriously, even before the ink dried on the label. Lilly has also flagged its next‑wave candidate retatrutide as a defining GLP‑1–based therapy, further entrenching its position in cardiometabolic disease.
Behind the scenes, Lilly is underwriting this future with heavy capital commitments, including a multi‑billion‑dollar manufacturing build‑out to support next‑generation injectable weight‑loss products. That kind of spend only makes sense if management believes the FDA is not just permitting, but structurally encouraging, rapid development of high‑impact metabolic drugs under accelerated frameworks. To investors used to thinking of the FDA as a speed bump, the GLP‑1 era is showing it can also act as a green light—at least for therapies that can move the needle on public‑health priorities like obesity, diabetes, and cardiovascular risk.
Merck: Oncology’s Master Class in Regulatory Endurance
Merck, meanwhile, is proving that while obesity may dominate headlines, oncology still sets the tempo for regulatory innovation. The company’s flagship checkpoint inhibitor, KEYTRUDA (pembrolizumab), continues to collect new indications and survival data, a trend reinforced by recent results in bladder and ovarian cancers showcased at a major European oncology congress. Positive survival data from KEYNOTE‑905 and KEYNOTE‑B96 are being highlighted in high‑profile sessions, underscoring how Merck uses pivotal trials not as a single swing at the plate but as a multi‑year indication expansion strategy.
At the same time, Merck is investing in next‑generation oncology assets, including antibody‑drug conjugates such as raludotatug deruxtecan (R‑DXd) for platinum‑resistant ovarian cancer. This kind of program is tailored for the FDA’s evolving stance on oncology approvals, where single pivotal trials, strong response data, and compelling survival curves can translate into meaningful, and sometimes expedited, regulatory decisions. In 2025 alone, the FDA cleared dozens of new drugs, with cancer therapies claiming a large share of the field, reflecting both scientific momentum and regulators’ willingness to move quickly when the clinical signal is compelling.
For Merck shareholders, the takeaway is that the company has become a case study in regulatory compounding: each successful trial, each label expansion, and each new mechanism doesn’t just add revenue; it builds a diversified reservoir of regulatory experience that can be redeployed across future oncology launches. In a sector where timelines and endpoints are as critical as molecules, that institutional memory is a competitive asset in its own right.
The New FDA Playbook: Speed for Those Who Earn It
On paper, the FDA approval process still begins with discovery and early research, winds through Phase 1 safety and Phase 2 proof‑of‑concept, and culminates in pivotal Phase 3 trials before regulators cast their vote. In reality, what’s changing is how that path is paved—through Fast Track and similar designations, pilot programs designed to compress review times, and a growing willingness to accept a single high‑quality pivotal study in certain diseases instead of the traditional two‑trial standard. For companies that can consistently deliver strong data against serious, high‑priority conditions, the regulator is increasingly acting more like a demanding partner than a distant referee.
Eupraxia is navigating that environment by focusing on precision delivery and leveraging designations like Fast Track to punch above its market‑cap weight. Eli Lilly is exploiting it at scale, turning its GLP‑1 engine into a platform for rapid launches and line extensions backed by expedited reviews and extraordinary demand. Merck, for its part, continues to demonstrate that rigorous oncology trials, thoughtfully sequenced and globally executed, can turn the traditional stepwise FDA diagram into a flywheel of recurring approvals.
For investors, the message is straightforward but not simple: the FDA is no longer a monolithic gatekeeper; it’s a dynamic system that rewards the right mix of innovation, safety, and execution. Companies that understand that—and build platforms, pipelines, and trial strategies accordingly—may find that the most valuable asset on their balance sheet is not any single drug, but their ability to navigate the increasingly intricate map hanging on that clinic wall.
The Sources
[1] 2025 FDA approvals – Nature https://www.nature.com/articles/d41573-026-00001-z
[2] Novel Drug Approvals for 2025 | FDA https://www.fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2025
[3] Eupraxia Pharmaceuticals Reports Positive Nine-Month Tissue … https://www.biospace.com/press-releases/eupraxia-pharmaceuticals-reports-positive-nine-month-tissue-health-and-symptom-data-from-the-highest-dose-cohort-in-its-ongoing-phase-1b-2a-resolve-trial-in-eosinophilic-esophagitis
[4] Eupraxia Pharmaceuticals https://eupraxiapharmaceuticals.com
[5] GLP-1 Pipeline Update: February 2026 – Prime Therapeutics – Portal https://www.primetherapeutics.com/glp-1-pipeline-update-february-2026
[6] Lilly’s Orforglipron, Retatrutide Named as Defining GLP-1 Drugs of … https://www.pharmexec.com/view/eli-lilly-orforglipron-retatrutide-glp-clarivate
[7] FDA Grants Speedy Approval to Eli Lilly’s Oral GLP-1 RA Drug for … https://carolinefifemd.com/2026/04/06/eli-lilly-oral-glp-1-ra-drug-for-obesity/
[8] Merck Advances Oncology Innovation, Highlighting Progress in New … https://www.merck.com/news/merck-advances-oncology-innovation-highlighting-progress-in-new-tumor-types-and-earlier-stages-of-disease-at-esmo-2025/
[9] Eli Lilly Uses GLP 1 Strength To Build Broader Drug Pipeline https://finance.yahoo.com/news/eli-lilly-uses-glp-1-210630652.html
[10] Eupraxia Pharmaceuticals to Present at Digestive Disease https://www.globenewswire.com/news-release/2026/04/22/3278788/0/en/eupraxia-pharmaceuticals-to-present-at-digestive-disease-week-annual-meeting.html
[11] Eupraxia Pharmaceuticals Receives U.S. FDA Fast Track … https://www.linkedin.com/pulse/eupraxia-pharmaceuticals-receives-us-fda
[12] Eupraxia to Present EP-104GI Data at DDW 2026 | EPRX SEC Filing https://www.stocktitan.net/sec-filings/EPRX/6-k-eupraxia-pharmaceuticals-inc-current-report-foreign-issuer-c59848386b1a.html
[13] Eupraxia Pharmaceuticals Receives U.S. FDA Fast Track … https://firstwordpharma.com/story/5750964
[14] The @fda approved 10 cancer drugs in 2025, based on clinical trials … https://www.facebook.com/memorialsloankettering/posts/the-fda-approved-10-cancer-drugs-in-2025-based-on-clinical-trials-in-which-msk-p/1283723167135231/
[15] IMG_9808.jpeg https://ppl-ai-file-upload.s3.amazonaws.com/web/direct-files/attachments/images/24996935/cb0c8d0f-ac5f-478b-86fc-f50961b36e2d/IMG_9808.jpeg?AWSAccessKeyId=ASIA2F3EMEYETJDLPFLC&Signature=rFhUy4Sb5m0bPaJJQlZZxcQqhKs%3D&x-amz-security-token=IQoJb3JpZ2luX2VjEJ3%2F%2F%2F%2F%2F%2F%2F%2F%2F%2FwEaCXVzLWVhc3QtMSJHMEUCIQDxoJG%2BCWewoD61vYlMwRdOUeW2eleqQznLispbOojWXgIgPQH4GaQJXHwbjjDWKCj5K8B%2FjGxHXI6iFdU1VJHlJmEq8wQIZhABGgw2OTk3NTMzMDk3MDUiDA%2Bo0bKMAqcu0CeDcSrQBKWx9JQ5Je8pEXdUTglNz4exZA1aUmft4VdElYabB%2BCGPl%2FB1XzDePDMe0KQrsJvyRJ%2BGroml8aiFSlJTFwaHu073hdCJx9G3j9TAUrMcnezLZv2RO4PgCbPIw1DSBTzgZtaRadIbWoe3STRFWdyOLmfVVdxjdUkOey3omGWvS7V6QqX2OFOFm2SpaQyTFHXYDZSw7fbiyV9Qqib9KCq4E6e03LeBqMBs2ZZ0utThgu6Uxmsey9h%2FD4Syoexb1v3waGz%2BY9pIGOXuSEDUM0Fa9qBJZSac%2B1V2oCREV%2BSvXpqH56LXj4jSGKXrXgKpZSejnG3mKM3aonmFUacYCCQuwYs3na3ouDoOrzQzjWMO1iFLS7%2BhCZ%2FsuRVEGczH5JfZBE7r9YIG8JP%2BpXa%2BVM8M9nAobYTFEGuGsFbZ%2BFhnxwbZIi9I2XXC%2Bq8ck01O%2BbJZSSZcNvpomtxNApPQcOrqszQFkHPBwOIGc6jDUo4vspviz6RY5cv%2FiEV060su%2BpZSS8VoCnFjoDd%2BJTPhHi7eVHIUOdEtQzkso4pYfdklgj%2F7DsIAr8m1Bk8vnTC2GGFZl9nbqWsEvevUb41uqDXy3wtcrkElMwVDlvotjEe0Avo61MXEFgAhVGmkmLJsOJ9Y8dvnSzB4%2FMrmjDjGR%2Fr6OAWbR7OM6ky3UMA%2BVwgT8EvAC1dO%2BcgC6DF69k9kbAO6V%2BMEaAe4SR6TkMBGlxaQCWmIyaMeo5w%2FHrhsG8SaWg3ZO%2BOxD1yMzGl5rDv5Qh5Rn0PNWN2hMlGEn00JlWNS7Ewrs7gzwY6mAHFv1hTVl0d0WaOo838PaBL4n5FdTOjrWX4H5s36r1PxueY1rj244l4uowCp4M6EboAoQ%2BzNnceq%2B26xpxKHJkD2IMJlv2eFw5Cs0hCDaQcnCKOpfbJGIhY%2BO4Fl3xVEQnqc2dj80QL0AfECtJ41DfoFKngLkHZScAVOgxBdSxk9fYlMSRs%2FouF7QKNy3uPAYVfqa183327Wg%3D%3D&Expires=1777872924
[16] Eli Lilly says new GLP-1 weight loss pill could reach market in 2026 https://www.youtube.com/watch?v=T_iLAFHEb9M


