
Eupraxia Pharmaceuticals (NASDAQ: EPRX) just earned a fresh vote of confidence from Raymond James, which is doubling down on its bullish thesis and arguing that the small-cap biotech now offers one of the most attractive risk/reward setups heading into 2026. The Raymond James update, issued on March 9, 2026, adds further institutional heft to a story that has been quietly building momentum.
A Strong-Buy Call With 2026 in Focus
Raymond James reiterates a Strong Buy rating on Eupraxia Pharmaceuticals (EPRX), highlighting the setup into Phase 2b RESOLVE data for lead asset EP‑104GI in eosinophilic esophagitis (EoE) expected in the second half of 2026. At a recent share price of 7.99 dollars and a market cap of roughly 405 million dollars, the firm argues that investors are still paying small‑cap prices for what could evolve into a much larger EoE franchise if upcoming clinical milestones break right.
EP‑104GI: A Differentiated Bet on EoE
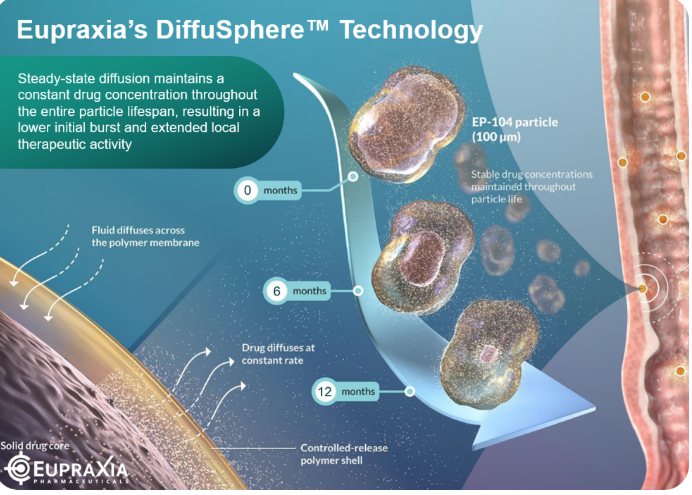
Raymond James frames EoE as a nascent, underpenetrated growth market with an estimated 3 to 5 billion dollars or more in peak U.S. addressable market potential alone. EP‑104GI, an extended‑release DiffuSphere formulation of fluticasone, has already posted encouraging open‑label efficacy in the RESOLVE Part A study, with limited systemic exposure and a once‑yearly administration concept that can be layered into routine endoscopy—an administration profile tailor‑made, as the analysts gently note, for gastroenterologists who prefer to be paid for their trouble.
Chasing Dupixent’s Neighborhood
In Raymond James’ view, the placebo‑controlled RESOLVE Part B study could deliver an efficacy profile that is roughly competitive with Sanofi’s (SNY) Dupixent, the current biologic weekly injectible heavyweight in EoE. If that thesis holds, the team sees room for more than a two‑times upside to its current net present value assumptions, suggesting that the market is treating Eupraxia like a prospect while the data package is quietly auditioning for the majors.
Financing Fuel and a “Pipeline-in-a-Product”
Eupraxia recently raised about 63 million dollars in equity financing, leaving the company well funded to deliver RESOLVE Part B topline results and move EP‑104GI into Phase 3 on positive data. The same capital also supports a broader gastrointestinal strategy, with planned expansion into fibrostenotic Crohn’s disease and esophageal strictures—indications the analysts describe as high‑need, high‑interest territories where a locally delivered, extended‑release steroid could evolve from single‑asset story to platform narrative.
Under-the-Radar Now, But Not Forever
Raymond James notes that, despite the recent launch of its coverage and a steadily rising price target history, EPRX and the RESOLVE Part B readout remain off the radar for many investors. Yet conversations with new shareholders suggest growing receptivity to the story, and the firm expects ongoing RESOLVE Part A updates through 2026 to help reframe Eupraxia from obscure Canadian clinical‑stage biotech to a name that portfolio managers feel compelled to have an opinion on—if not a position in.
Risks Still Matter – This Is Biotech, After All
The report is careful to remind readers that this is classic aggressive‑risk biotech territory, with the usual suspects in play: clinical and regulatory uncertainty, commercial and competitive pressure, intellectual‑property considerations, and the possibility of future financing needs if the development timeline stretches. Still, within that high‑beta universe, Raymond James’ analysts position Eupraxia as one of their better calibrated risk/reward ideas for 2026, arguing that Part A data have already de‑risked key aspects of the program and left meaningful upside tied to a single, highly visible catalyst.
Who Wrote the Report – And How to Reach Them
The Eupraxia report was authored by lead analyst Martin Auster, MD, with contributions from Thomas Deal, CPA (Senior Research Associate), Joshua Yan (Research Associate), and Jakob Hebert, PhD (Research Associate). Institutional investors typically can request the full report or engage with the Raymond James team by contacting their Raymond James sales representative or relationship manager, or by reaching out through the firm’s published research contact channels listed on the report cover or the Raymond James institutional research portal.
The Sources
- Raymond James – “Eupraxia Pharmaceuticals Inc. (EPRX): Following Up Post Launch: We See an Attractive Setup for 2026 Into Ph2b RESOLVE EoE Data,” Global Research report, March 9, 2026
(Access typically via Raymond James institutional research platforms or your Raymond James representative: https://www.raymondjames.com) - Raymond James – Legal disclosures, methodology, rating definitions, and global distribution notices (embedded in the Eupraxia Global Research report, pages 2–9).



